Cancer isn’t on your terms, but the care you get should be
Get startedWe are a virtual care clinic
Connecting people living with cancer, and their caregivers, to a team of multidisciplinary cancer experts
Get started
Better outcomes and lower costs for everyone
We provide 1-to-1 appointments with NHS-registered cancer specialists and expert-led content for every stage of the journey
Get started

On-demand access to the UK’s largest collective of cancer experts
Get startedMultidisciplinary care as standard
Get started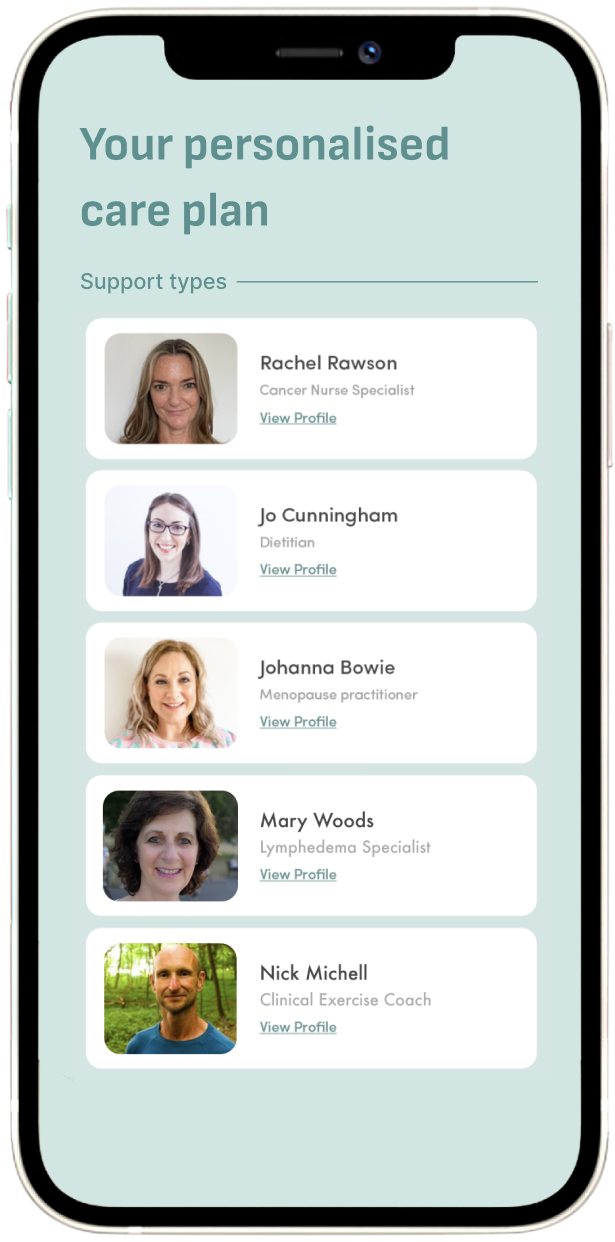
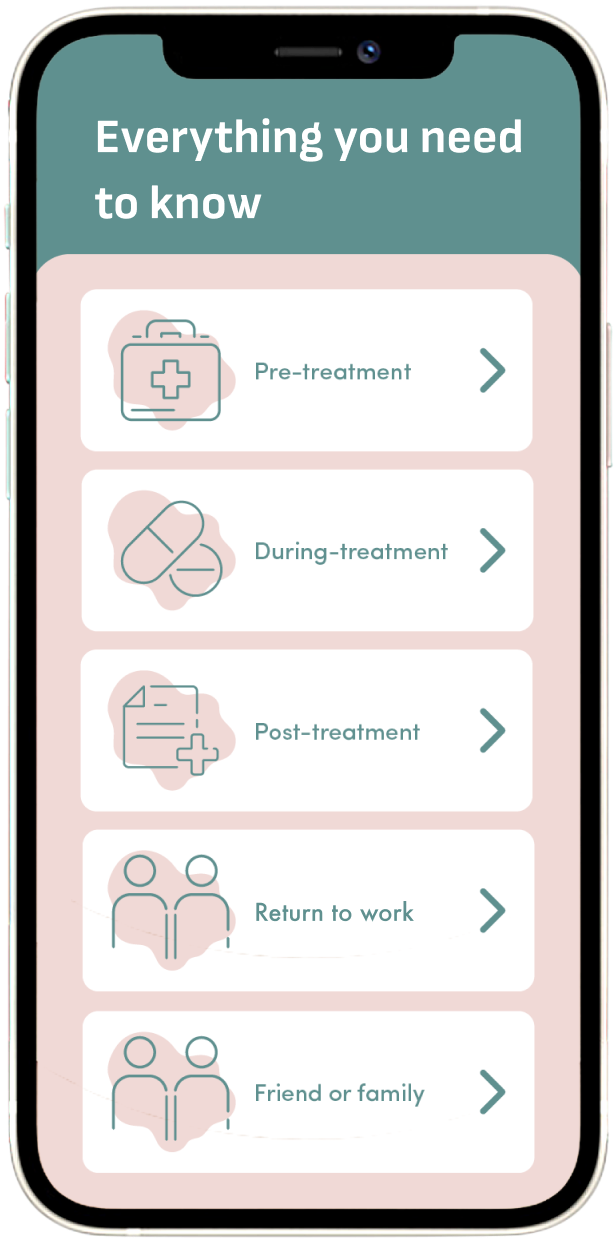
Connected support through every phase, all on one digital platform
Get started
Comfort
No question is too big or too small. Our healthcare professionals are here to listen and offer guidance, whenever you need it.
Get started
Convenience
See a cancer expert from home. Perci simplifies the process of getting care that is created for you and your needs.
Get started
Collaboration
Working with your GP and oncology team, we deliver care that connects the dots
Get started
Credibility
Our cancer experts include NHS-trained cancer nurses, registered dietitians, psychologists, physiotherapists, menopause practitioners and many more
Get started